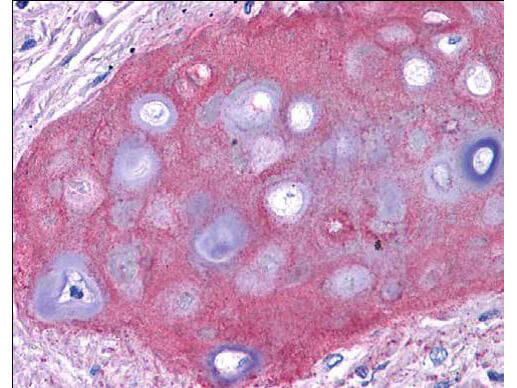
What Are Collagens? Understanding the Basics
Collagens are essential components of the extracellular matrix and continue to be important targets for technology and therapy. As the chief structural component of skin, collagen is the most abundant protein in the body, representing approximately 30% of its dry weight. The use of collagen has become increasingly prevalent in various medical applications such as implants, organ replacement, tissue equivalents, arterial plugs, cosmetic surgery, surgical sutures, and surgical dressings for wounds and burns. These advances were predicated on the use of anti-collagen antibodies to allow researchers to detect collagens, including type-specific collagens, in cell biology research.
Illustration depicting the triple helical structure of collagen
Variations in collagen structure create functional diversity essential for distinct biological features in the various types of tissues of the body. Based on their supramolecular structures, collagens are divided into two main classes: fibril-forming collagens (type I, II, III, and V) and non-fibril-forming collagens (type IV and VI). In humans, there are more than 20 unique procollagens that are subjected to various post-translational modifications resulting in great diversity between collagen types.
Collagen Antibodies
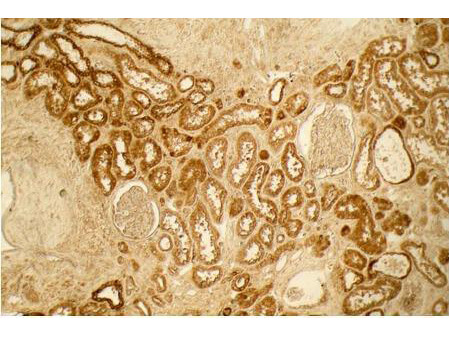
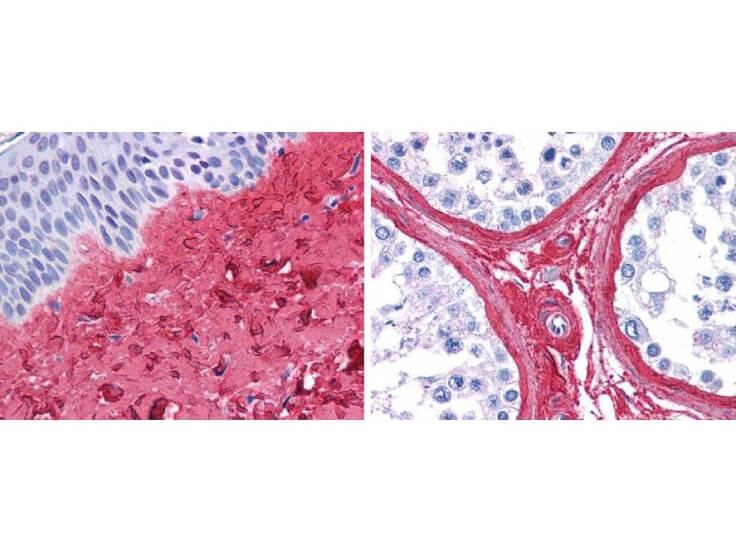
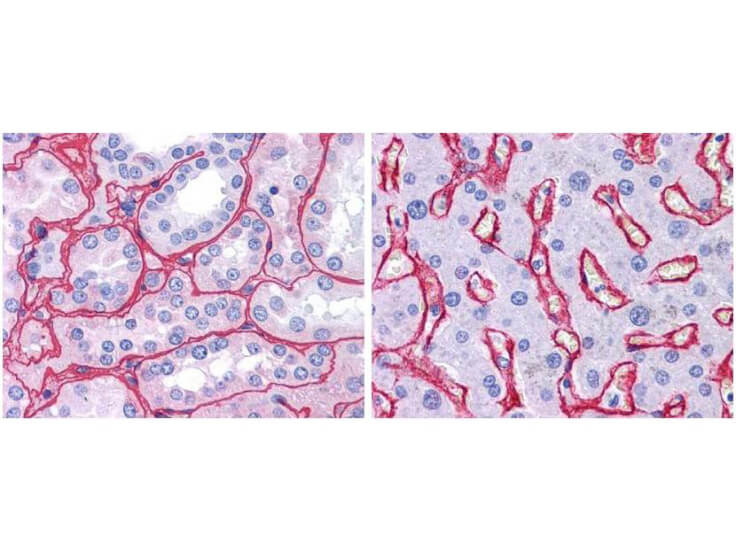
Precise insights into the behavior and physiology of cells can be made by researchers, especially when using a type-specific collagen antibody to study collagen’s bio-distribution, interactions between associated cells, mechanisms of cell adhesion, and cell development and differentiation. Because of the high specificity of type-specific anti-collagen antibodies, which often differentiate one collagen type from another by subtle differences in the triple-helical structure of native collagen molecules, it is important to select the correct antibody that is “fit-for-purpose” to detect a specific type of collagen in the right tissue and immunoassay. Rocklands collagen antibodies have been cited in more than 300 research publications, establishing them as the preferred option in nearly every circumstance when it comes to collagen research.
Collagen Antibodies
| Product | Clonality | Reactivity | Applications |
| Collagen Type I Antibody | Polyclonal | Human, Mouse, Rat, Bovine, Pig | WB, IHC, IF, FC, IP, Multiplex, ELISA |
| Collagen Type II Antibody | Polyclonal | Human, Bovine | IHC, IF |
| Collagen Type III Antibody | Polyclonal | Human, Bovine, Pig | WB, IHC, IF, FC, ELISA |
| Collagen Type IV Antibody | Polyclonal | Human, Bovine | WB, IHC, IF, Multiplex |
| Collagen Type V Antibody | Polyclonal | Human, Bovine | IHC, IF, Multiplex |
| Collagen Type VI Antibody | Polyclonal | Human, Bovine | IHC, IF |
Collagen Antibody Controls
It cannot be understated that the appropriate controls should be included in experimental design when type-specific anti-collagen antibodies are used to ensure the success of the experiment and data reproducibility. Only highly-purified and type-specific collagen proteins should be used as controls that are themselves free of impurities, including serum proteins, other collagen proteins, and non-collagen extracellular matrix proteins. Standards must not only be physically pure for collagen but also be immunologically pure for the type of collagen to be used as controls in experiments of this type. Type-specific anti-collagen antibodies should be selected that are appropriate for the detection of collagens present in the tissue to be tested, and these antibodies should be used with protocols designed to optimize antibody performance for the collection of high-quality data ultimately intended to help better understand the biology of these proteins.
Human Collagen Proteins
| Product | Origin | Applications |
| Human Collagen Type I | Human | SDS-PAGE, WB, Cellular Assay |
| Human Collagen Type II | Human | SDS-PAGE, WB |
| Human Collagen Type III | Human | SDS-PAGE, WB |
| Human Collagen Type IV | Human | SDS-PAGE, ELISA |
| Human Collagen Type V | Human | SDS-PAGE |
| Human Collagen Type VI | Human | SDS-PAGE, Cellular Assay |
Bovine Collagen Proteins
| Product | Origin | Applications |
| Bovine Collagen Type I | Bovine | SDS-PAGE, WB |
| Bovine Collagen Type II | Bovine | SDS-PAGE |
| Bovine Collagen Type III | Bovine | SDS-PAGE |
| Bovine Collagen Type IV | Bovine | SDS-PAGE |
| Bovine Collagen Type V | Bovine | SDS-PAGE |
| Bovine Collagen Type VI | Bovine |
Top Cited Collagen Antibodies
Frequently Asked Questions