Immunohistochemistry Technique
Immunohistochemistry (IHC) is a mainstay immunoassay for many laboratories and is used to demonstrate the presence or absence of important proteins, detect post-translational modifications, diagnose disease, and more. Rockland produces highly-active antibodies and conjugates for use in IHC experiments. Rockland also produces high-quality reagents for the setup and detection in IHC experiments. Protect your experiments with Rockland antibodies.
IHC uses both immunological and biochemical techniques to examine distinct tissue sections by the use of labeled antibodies interacting with target antigens. A visible label or stain is used to visualize the antibody antigen interaction. IHC allows for the visualization of distinctive cellular components of cells and is frequently used by pathologists and in diagnostics.
IHC Sample Preparation
The antigen is fixed and adhered to a glass microscope slide. Formaldehyde fixation is suggested as the routine initial method of choice for tissue and cell fixation, but other methods are also suitable. Background staining is reduced with a blocking buffer to block non-specific sites to which the antibodies may bind. Rockland offers common blocking buffers including BSA, normal serum, non-fat dry milk, and an optimized proprietary blocking buffer.
IHC Sample Labeling
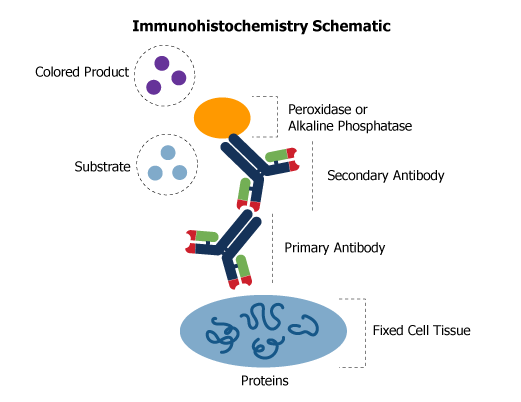
Labeling is achieved by the reaction of a primary antibody with the immobilized antigen to form an antigen-antibody complex. A second biotinylated antibody specific for the primary antibody host species reacts with the complex. Streptavidin conjugated to the enzyme peroxidase or alkaline phosphatase reacts with this complex. Finally, a substrate is added to causes a reaction with the enzyme, producing a colored precipitate to form on the slide at the location of the antigen. The slide is viewed through a standard light microscope and photographed. A common manipulation in IHC fixing is the use of the antigen retrieval technique, a simple method of heating or boiling paraffin-embedded tissue sections to enhance the observed signal.
Immunohistochemistry Kits

The MaxTag™ Histo Immunohistochemistry Kits allow for the detection of primary polyclonal or monoclonal antibodies provided by the user. The kits rely on the high-specificity of avidin-biotin binding for visualization of an antigen and are intended to provide a simple, reliable, and convenient detection system for immunohistochemical staining. The kits include all regents required for IHC, such as an antibody, buffers and substrates, fixative, and counterstain. These simple-to-use kits guide you through IHC using highly optimized procedures that result in low background and high signal, producing the most sensitive detection kit available.