Product Categories
Primary Antibodies(4672)
Polyclonal Antibodies(3770)
Cell Biology(2134)
Cancer(1787)
Neuroscience(1066)
Cell Signaling Pathways(1049)
Monoclonal Antibodies(889)
Immunology(642)
Epitope-Tag Antibodies(311)
COVID-19(232)
Epigenetics(207)
Microbiology(158)
RFP and GFP Antibodies(42)
Loading Control Antibodies(24)
Recombinant Antibodies(12)
Secondary Antibodies(2277)
Conjugated Antibodies(1906)
Pre-adsorbed Antibodies(678)
Fab Fragment Antibodies(536)
Unconjugated Antibodies(389)
Proteins and Peptides(1485)
Other(504)
Immunoglobulins(378)
Cytokines and Growth Factors(316)
Enzymes(170)
Isotype Controls(70)
Transferrins and Albumins(26)
Protein A and G(25)
Collagens(20)
Kits(858)
ELISA Kits(732)
Other(72)
Western Blot Kits(39)
IP Kits(14)
Cell Viability Kits(4)
Cell Lines and Lysates(627)
Cell Lines(372)
Animal Tissues(165)
Cell Lysates(90)
Blood Products(513)
Serum and Plasma(405)
Whole Blood(37)
Tissue Powders(25)
Complement(17)
Gamma Globulin Fractions(15)
Red Blood Cells(14)
Supporting Reagents(173)
Molecular Biology Reagents(73)
Western Blot Reagents(53)
Avidin, Biotin, Streptavidin(41)
Blocking Buffers(28)
ELISA Reagents(25)
Assay Substrates(16)
TrueBlot(26)
TrueBlot IP Beads(15)
TrueBlot Secondary Antibodies(11)
TrueBlot Kits(8)
Resource Type
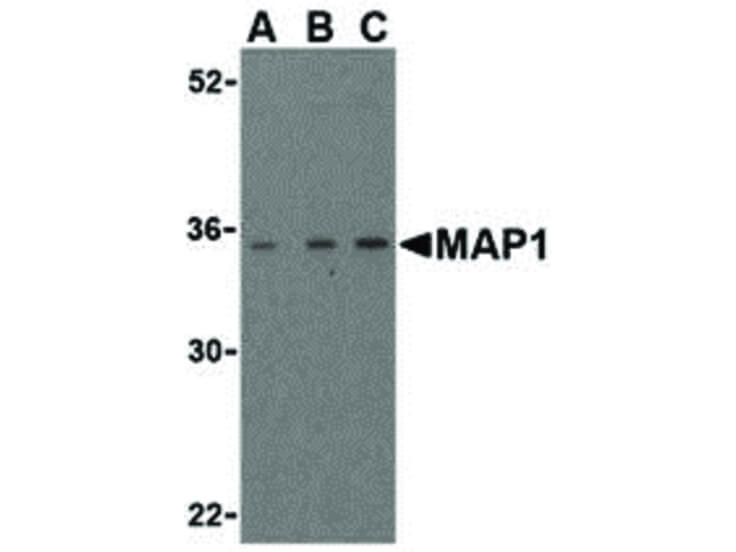

-HRP-1-E-4x3.jpg)