VEGF-A Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
VEGF is a potent mitogen in embryonic and somatic angiogenesis with specificity for vascular endothelial cells. VEGF forms homodimers and exists in four different isoforms. Overall, the VEGF monomer resembles that of PDGF, but its N-terminal segment is helical rather than extended. VEGF shares homologies of about 21% and 24% respectively with the A and B chains of human platelet-derived growth factor (PDGF), and has complete conservation of the eight cysteine residues found in both mature PDGF chains. The cysteine knot motif is a common feature of this domain. The homology is not reflected in function, however, since the cell types responsive to VEGF are distinct from those responsive to homo- and heterodimers of the PDGF chains. This protein is a glycosylated mitogen that acts on endothelial cells and has various effects, including mediating increased vascular permeability, inducing angiogenesis, vasculogenesis and endothelial cell growth, promoting cell migration, and inhibiting apoptosis. VEGF-A also has been shown to have effects on a number of other cell types (e.g. stimulation of monocyte/macrophage migration, neurons, cancer cells, kidney epithelial cells ). VEGF-A is also a vasodilator; it increases microvascular permeability, and was originally referred to as vascular permeability factor. Alternatively spliced transcript variants, encoding either freely secreted or cell-associated isoforms, have been characterized.
Product Details
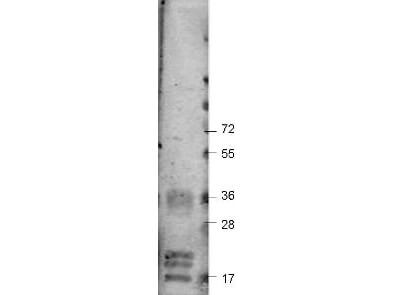
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.