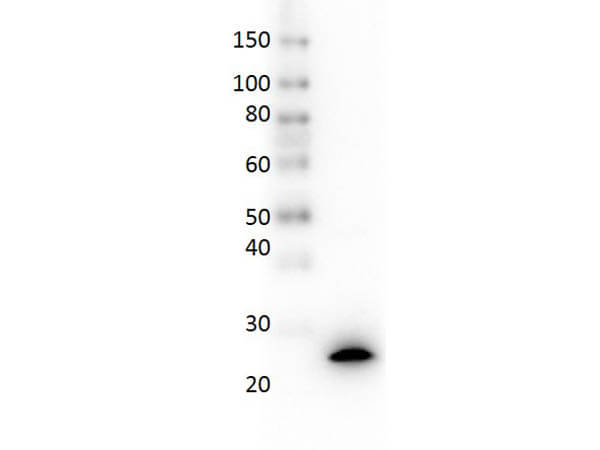
IL-35 EBI3 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
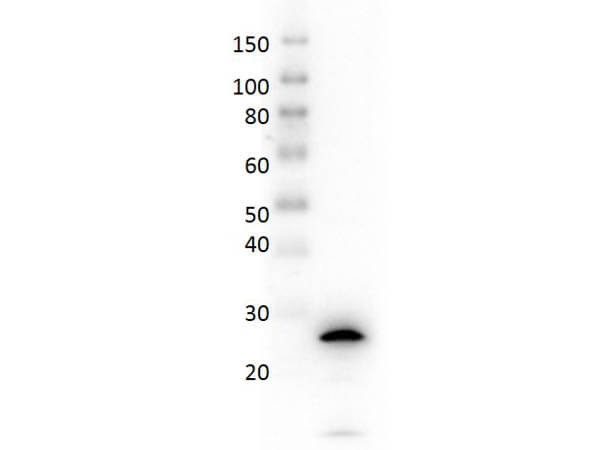
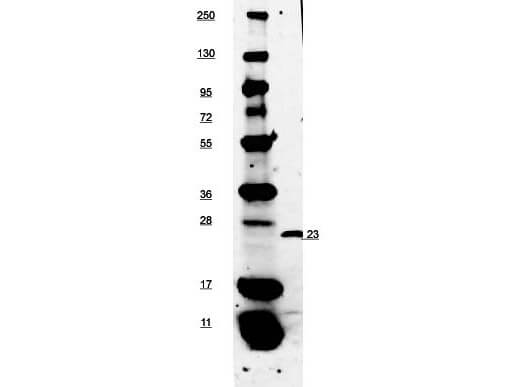
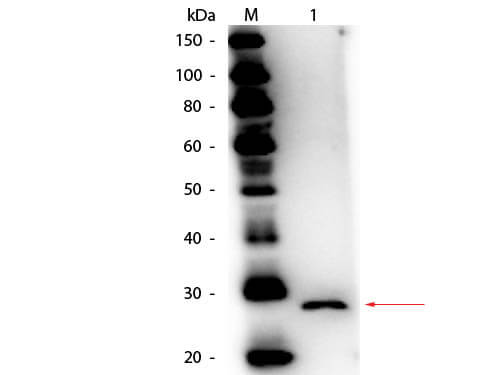
The cytokine Interleukin 27 (IL-27) is produced in response to inflammation. It is made by activated antigen presenting cells including monocytes, endothelial cells, and dendritic cells. IL-27 consists of a heterodimeric combination of Epstein-Barr virus-induced molecule 3 (EBI3, or IL-27B) non-covalently linked with IL-27 p28 (or IL-27A). It is a regulator of T helper cell development and suppressor of T-cell proliferation. IL-27 has both pro- and anti-inflammatory properties. It can stimulate cytotoxic T cell activity and induce isotype switching in B-cells. It has diverse effects on innate immune cells. It induces monocytes and mast cells to secrete pro-inflammatory cytokines. When infection is present, IL-27 induces naive CD4+ T cells to proliferate and develop Th1 cell responses. As an anti-inflammatory regulator, IL-27 can inhibit Th1 or Th2 responses and restrict the strength and duration of adaptive immune responses. The IL-27 p28 subunit, a 28 kDa glycoprotein belonging to the type I cytokine family, is homologous to IL-12 p35, IL-23 p19, and IL-6. The EBI3 (Epstein-Barr virus-induced molecule 3, or IL-27B) subunit is a 34 kDa glycoprotein containing two fibronectin type III domains, and belongs to the type I cytokine receptor family. It can exist as a homodimer and can also heterodimerize with IL-12 p35. It is homologous to the p40 subunit of IL-12 and IL-23 and to the extracellular domain of IL-6 R. EBI3 can heterodimerize also with IL-12 p35, or can exist as a homodimer.
Product Details
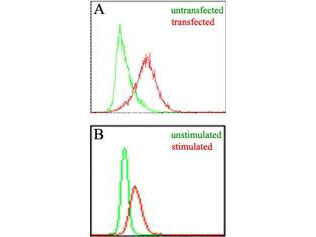
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.