FIV matrix protein p15 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
This antibody is designed, produced, and validated as part of a collaboration between Rockland and the National Cancer Institute (NCI) and is suitable for Cancer, Immunology and Nuclear Signaling research. Feline immunodeficiency virus (FIV) belongs to the lentivirus family. This family is characterized by assembly of the viral capsid at either the plasma membrane or at the limiting membrane of late endosomes. The capsid assembles from the viral Gag polyprotein. Upon release of a budding virion, Gag precursor protein is cleaved by the viral protease into its mature products, namely Matrix Protein, Capsid and Nucleocapsid. Matrix Protein, located at the N-terminus of the Gag polyprotein, is usually myristylated during protein translation, prior to the later events of virus assembly. The myristate moiety is believed to be sequestered within the Matrix Protein during protein translation and later facilitates membrane binding upon exposure resulting from conformational changes. Essential functions attributed to the Matrix Protein of lentiviruses include targeting newly synthesized Gag precursor proteins to the site of virus assembly by binding with cellular components such as phosphatidylinositides. In the mature virus particle, the Matrix Protein provides internal structure to the virion within the capsid, but is not exposed at the surface of the particle. Based on studies with HIV, it is postulated that FIV Matrix Protein may also serve additional functions, including nuclear localization of the viral core upon entry of the virus into a new host cell.
Product Details
Target Details
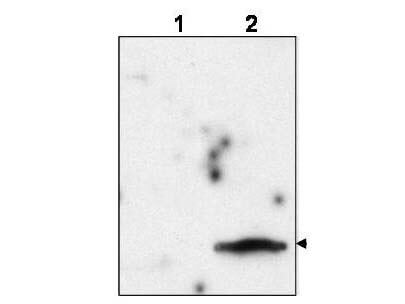
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.