Beta Amyloid 9 Antibody
Sheep Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
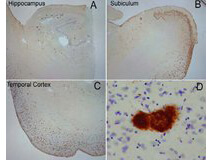
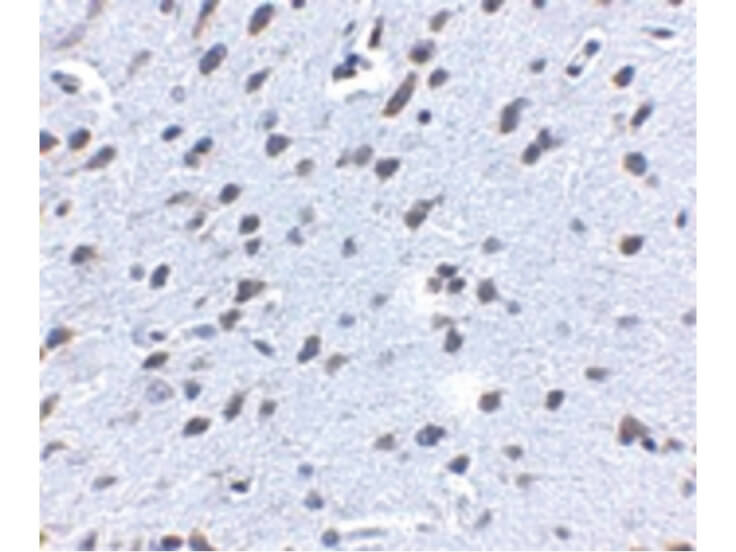
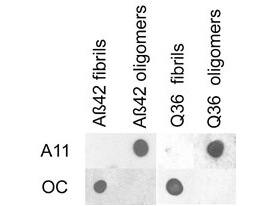
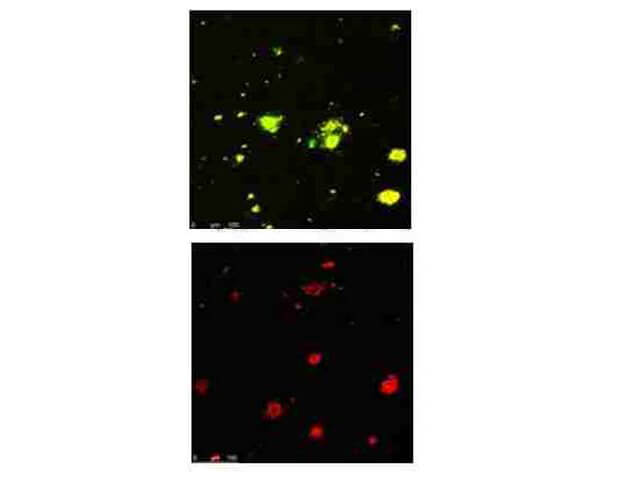
Beta Amyloid functions as a cell surface receptor and performs physiological functions on the surface of neurons relevant to neurite growth, neuronal adhesion and axonogenesis. It is expressed in all fetal tissues with the highest levels located in the brain, kidney, heart and spleen tissue. It is involved in cell mobility, transcription regulation via protein-protein interactions and copper homeostasis/oxidative stress through copper ion reduction. The copper-metallated APP induces neuronal death directly in vitro, or is potentiated through Cu2+ mediated low-density lipoprotein oxidation. It has binding capabilities via its C-terminus for transient metals such as copper, zinc and iron. It binds APBB1-KAT5 to promote transcription activation and inhibits Notch signaling through interaction with Numb. It also promotes tau aggregation and TPK II-mediated phosphorylation. Anti-Beta Amyloid regulates neurite outgrowth by binding components in the cellular matrix such as heparin, collagen I and amyloid-beta peptide, leading to mitochondrial dysfunction in cultured cortical neurons. Defects in APP cause Alzheimer disease type 1 and cerebral amyloid angiopathy. This antibody is ideal for researchers interested in Cancer or Neuroscience research.
Product Details
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.