HSP90 K294 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
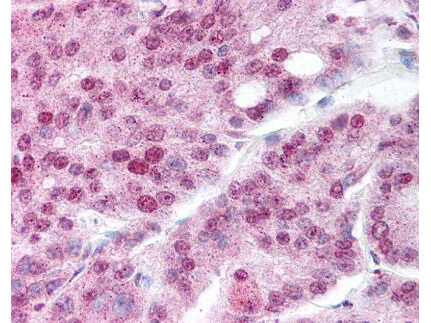
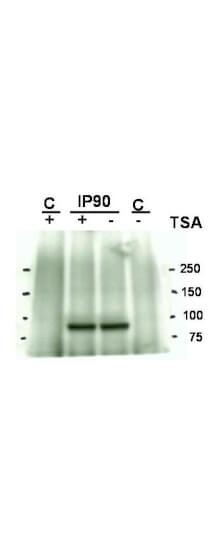
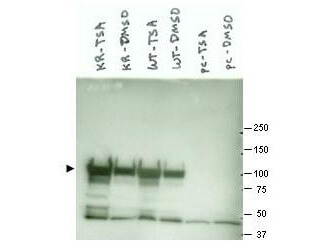
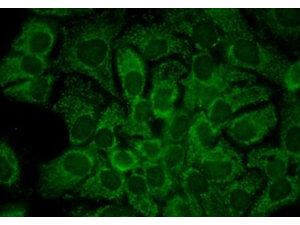
This antibody is designed, produced, and validated as part of a collaboration between Rockland and the National Cancer Institute (NCI) and is suitable for Cancer, Immunology and Nuclear Signaling research. Hsp90 is a member of the heat shock protein 90 family, the members of which are highly conserved between isoforms and species. Hsp90 functions as a molecular chaperone and has ATPase activity. Hsp90 is a cytoplasmic protein that forms a homodimer in vivo and interacts with TOM34, AHSA1, HDAC6 and SMYD3. Several signal transduction pathways depend on Hsp90 function, including pathways involving erbB2, hypoxia sensitivity (Hif1 alpha), and steroid hormone receptors (for example, androgen, progesterone, glucocorticoid, and aryl-hydrocarbon). Recent reports show that Hsp90 from tumor cells has increased sensitivity to small molecule inhibitors (for example, 17AAG). The mechanism of the differential sensitivity of Hsp90 from normal versus tumor cells is unknown, although mutation has been ruled out. One possible mechanism may be differences in post-translational modification of tumor Hsp90. K294 was found to be acetylated in purified Hsp90 from SkBr3 cells, a breast cancer cell line.
Product Details
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.