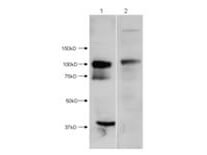
HDAC7 (N-terminus) Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
HDAC7 is a member of the class II mammalian histone deacetylases, which plays an important role in modulating the eukaryotic chromatin structure. Human HDAC7 is composed of 912 amino acid residues. Although HDAC7 is localized mostly to the cell nucleus, it is also found in the cytoplasm, suggesting nucleo-cytoplasmic shuttling. The histone deacetylase activity of HDAC7 maps to a carboxy-terminal domain and is dependent on interaction with class I HDACs in the nucleus. It is an active component of different transcriptional corepressor complexes that can be recruited to specific promoter regions via interactions with a growing number of sequence specific transcriptional factors. HDAC7 catalyzes removal of acetyl-groups from acetyl-lysines of histones and promotes compaction of chromatin in these regions, leading to the inhibition of gene transcription. Anti-HDAC7 antibodies are ideal for researches interested in Breast Cancer, Cancer, Cell Cycle and Replication, Chromatin Research, Epigenetics, and Histone Deacetylases research.
Product Details
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.